Experiment - Conductors and insulators
In this experiment you will make observations on the ability of different materials to conduct electricity and gain an insight into the meaning of conductor and insulator.
Aim |
To examine the nature of conductors and insulators
Make a table listing all of the substances you intend to test to see if the substance is a conductor or insulator. Make a column headed prediction then another column indicating whether the substance is an insulator or a conductor. Write down whether you think it will conduct electricity before you perform your test then perform the test to check whether your prediction is correct or incorrect. |
Apparatus (Equipment) |
1 x DC variable power supply, with a nominal range from 2 to 12 V.
1 x 6V 10 W light globe
2 x conducting leads with alligator clips
2 x conducting leads with suitable attachments for battery or power pack
1 x tapping key switch
Various materials to test between contacts, e.g. wood, paper, plastic, alfoil, copper plate, a nail, glass sheet, Perspex rod, etc. |
Method |
The circuit below was setup : 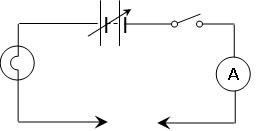
Label all the symbols used on the circuit diagram. Why is there a break in the circuit?
The variable DC power supply was set to the 6 V setting.
The different test materials were, one at a time, attached between the contacts.
The power was then switched ON and careful observations on the appearance of the light globe were made and any reading on the ammeter was noted.
The observations were recorded.
This was repeated for each of the test materials.
The results were recorded, analysed, and then a conclusion written.
|
Questions
- What did you observe when the test material was a GOOD conductor?
- Which of the materials was an insulator? How did you know?
- Based on your observations, can you predict which type of materials are normally good conductors and which materials are normally good insulators?
- What is suggested about the electrons in the chemical bonds of a material if the material is a GOOD insulator?
- Metals are said to have a cloud of loosely held electrons that enables them to have properties of being good conductors. What observations made by you support the idea that the metals have a cloud of loosely held electrons that relatively, are free to move?
